Advanced Platform Technology Center
Micropackage Technology for Implantable Microsystems
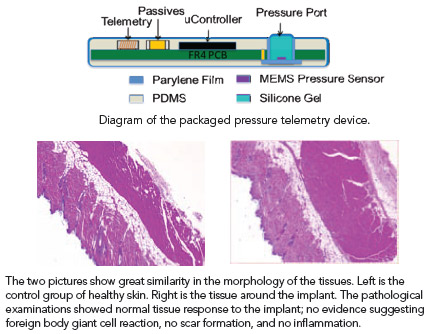
Implanted medical devices containing active electronics currently need to be hermetically sealed in titanium or ceramic packages, which limit their size, mechanical flexibility and integration with biological tissues. Investigators with the APT Center are developing unique nonhermetic (not airtight), biocompatible micropackaging approaches based on the engineering of novel multilayer materials and thin film coatings.
The encapsulation we are developing is typically thinner than 250 µm (micromilimeters) and adds minimal volume to the system being packaged. The multilayer thin film approach allows flexible wires and electrode arrays to be inserted within the same thin flexible protective coating as any rigid integrated circuits and components to eliminate the weak points of traditional feed-throughs and cable connections. Our approach also produces soft, smooth outer layer that is mechanically and biochemically compatible with the tissue and may reduce the potential for irritation or rejection responses with movement of application of local pressure.
The non-hermetic micropackage technology we have developed thus far exhibits an accelerated life time of up to 5 years. Biocompatibility testing is similarly encouraging with all animals surviving the one month implantation period with no acute histological problems or pathologies observed at explant.
Principal Investigator: Wen H. Ko, Ph.D.
Clinical Collaborator: Margot Damaser, Ph.D.
Funding Agency: National Institutes of Health